- 25 Feb 2025
- 6 Minutes to read
Simulating battery fires
- Updated on 25 Feb 2025
- 6 Minutes to read
Battery Energy Storage System (BESS) thermal runaway
Batteries can pose significant hazards, such as toxic and flammable gas release, fires and explosions, which can harm people and damage assets. Once a battery fire starts, it can be extremely difficult to control. Even if the flames are extinguished, reactions within the batteries can continue, generating flammable gas that may reignite. Another consequence of battery fires is the release of toxic gases such as hydrogen fluoride, which can disperse into the surrounding area. Hydrogen fluoride is a toxic chemical that can cause harm even at low concentrations.
Scenario
Toxic gas release from thermal runaway of BESS system | |
|---|---|
Uncontrolled exothermal reaction in individual cells leads to thermal runaway. Heat propagation to adjacent cells generates toxic and flammable gases. In this scenario, toxic gas production and dispersion from a partial and full BESS thermal runaway will be modelled.
| |
Partial BESS thermal runaway Thermal runaway propagation is limited to one module. Heat release is limited and gas could cool down within the system before dispersing. The release can last for less than an hour. | Full BESS thermal runaway Thermal runaway propagation reaches the total BESS (full fire). Heat release is high, but ambiguous to calculate. The release can last hours - days. |
The BESS thermal runaway event tree below provides a detailed analysis, specifically outlining hazards arising from battery damage due to thermal, electrical, or mechanical abuse. Using this event tree, we can assess the possible events arising from a BESS thermal runaway.
Flash fires: Flammable gases like hydrogen and hydrocarbons can ignite, causing flash fires.
Vapour cloud explosions (VCE): Flammable clouds encountering congested areas and confined spaces, may lead to vapour cloud explosions.
Toxic cloud: Battery material decomposition releases toxic gases, including HF, HCl, HCN, CO, and NO₂. Hydrogen fluoride (HF) is particularly concerning due to its high toxicity and corrosive properties.
.png)
BESS thermal runaway event tree
Please note that the EFFECTS battery model can only assess the production of toxic gases from BESS thermal runaway, allowing for the modelling of toxic dispersion. Flammable scenarios are outside the model's application range.
Modelling approach
To simulate this scenario, you can follow the following steps:
Add background. Define the context and environment for the simulation.
Add receivers (optional). Identify vulnerable areas where toxic cloud dispersion might occur.
Add equipment. Set the location of the ammonia release takes place on the map.
Select models. Start from the point of release and add subsequent models in the order of events to create a model tree.
Model selection
At the moment EFFECTS focuses only on toxic gas formation from BESS thermal runaway and its dispersion while the flammable gas formation or heat propagation is not taken into account. Out of the produced gas mixture specifically, hydrogen fluoride presents unique challenges, as it may form heavier oligomers or react with water vapour, generating heat and becoming lighter. The ‘‘Dispersion’’ model in EFFECTS does not account for these phenomena. That is why the neutral gas dispersion model is recommended for modelling dispersion from BESS releases providing a rather conservative but reliable estimate, accounting for these complexities in a safe manner. Additionaly neutral gas dispersion model:
Includes Lee-Wake behaviour to assess predilution behind BESS.
Neglects buoyancy due to heat production from full BESS thermal runaway which is conservative, but currently often done in practice.
The toxic cloud dispersion is modelled using the ‘‘Neutral gas - Toxic dose’’ model. To determine inputs for this model, the ‘‘Li-ion Battery Storage Thermal Runaway’’ release model is used upfront to estimate the source term by calculating HF, CO, HCl, HCN, and NO2 formation rates and released mass in time.
Scenario definition:
Partial BESS Thermal Runaway
Safety measures (e.g. spacing between modules) confine thermal runaway to a single module (80 kWh). Thermal runaway over 30 min.
Full BESS Thermal Runaway
Full BESS reaches thermal runaway over 12 hours. Such estimation is more in line with the duration of full BESS fires, which can last for days.
The simulation starts with the release model ‘‘Li-ion Battery Storage Thermal Runaway’’. This model in EFFECTS requires several inputs:
Battery chemistry: either NMC or LFP, as their characteristics will affect the release rate of toxic substances.
The cell's electrical energy capacity represents the energy content of the battery cells involved in the thermal runaway event. The capacity can be defined per modules/cells or the entire system.
The duration of the release is the total time over which the thermal runaway and gas release occur.
The level of conservatism is an expert parameter that can be set to different levels from low to high.
Full BESS fire I n 12 hours | Single module TR in 30 mins | |
Level of conservatism | High (95% percentile) | High (95% percentile |
|---|---|---|
Source Definition | ||
Battery chemistry | LFP | LFP |
Energy capacity based on | Battery system | Battery module |
Energy capacity (kWh) | 3500 | 80 |
Number of modules | - | 1 |
Duration of the release (s) | 43200 | 1800 |
After the release model provides formation rates of HF, CO, HCl, HCN, and NO2, each of these rates can be connected to a dispersion model. Hydrogen fluoride (HF) is particularly concerning due to its high toxicity, therefore is selected for further dispersion calculation. The neutral gas model allows release definition from the user-defined window which in this case represents the battery system (the container is 2.3 m in width and height). The release will take place at 1.15 m height.
User-define window dimensions in ‘‘Neutral gas’’ dispersion model
Other additional parameters that need to be defined are presented in the table below. These include the definition of release type, meteorological and environmental conditions, and vulnerability settings defining the translation from physical effects to damage.
The meteorological and environmental conditions will influence and determine turbulence in the atmosphere.
Toxic exposure duration is based on a defined approach of applying exposure limit. This can be limited by the time needed for sheltering, delayed by the cloud’s arrival at the location or limited by release duration.
Process Conditions | Full BESS fire in 12 hours | Single module TR in 30 mins |
Chemical name | HYDROGEN FLUORIDE (DIPPR) | HYDROGEN FLUORIDE (DIPPR) |
|---|---|---|
Source Definition | ||
Type of neutral gas release | Semi-continuous | Semi-continuous |
Type of continuous source | User defined window | User defined window |
Mass flow rate of the source (kg/s) | 0,018095 | 0,0099263 |
Duration of the release (s) | 43200 | 1800 |
Length source in wind (x) direction (m) | 0 | 0 |
Width source in crosswind (y) direction (m) | 2,3 | 2,3 |
Height source in vertical (z) direction (m) | 2,3 | 2,3 |
Process Dimension | ||
Height of release (Z-coordinate) (m) | 1,15 | 1,15 |
Offset X direction (distance) start dispersion (m) | 0 | 0 |
Offset Z direction (height) start dispersion (m) | 0 | 0 |
Roughness length description | Open flat terrain; grass, few isolated objects. | Open flat terrain; grass, few isolated objects. |
Environment | ||
Ambient temperature (°C) | 9 | 9 |
Ambient pressure (bar) | 1,0151 | 1,0151 |
North/South latitude of the location (deg) | 51 | 51 |
Roughness length description | High crops; scattered large objects, 15 < x/h < 20. | High crops; scattered large objects, 15 < x/h < 20. |
Vulnerability | ||
Toxic exposure duration based on | Time limit until sheltering | Time limit until sheltering |
Start of exposure (after moment of release) (s) | 0 | 0 |
Max. duration until sheltering (s) | 1800 | 1800 |
Results
EFFECTS can be used to analyse concentration levels and dose accumulated both outdoors and indoors, while also presenting contours for various standard concentration thresholds. In this case, we are interested in ERPG-1, ERPG-2, and ERPG-3 distances. The displayed thresholds can be selected by the user in the presentation settings node of the EFFECTS project tree.
.png)
Dispersion model concentration results for 12-hour release from BESS system - ERPG-1, ERPG-2, and ERPG-3 contours
In addition to the map view, the graph tab displays concentration levels over distance and time, a side view of plumes for selected thresholds, and lethality and dose data over distance from the source, as shown below for the full BESS thermal runaway scenario.
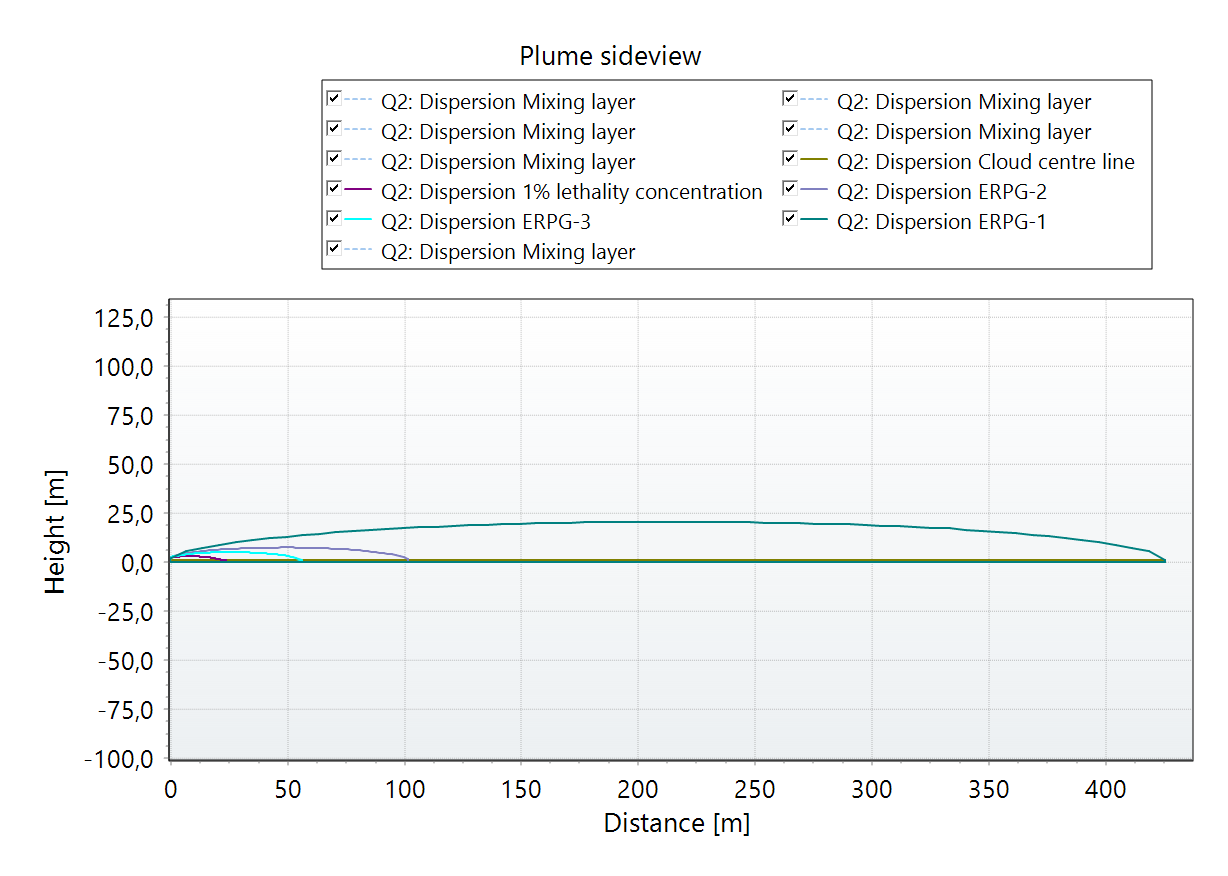 Toxic dispersion of HF - plume side view. | 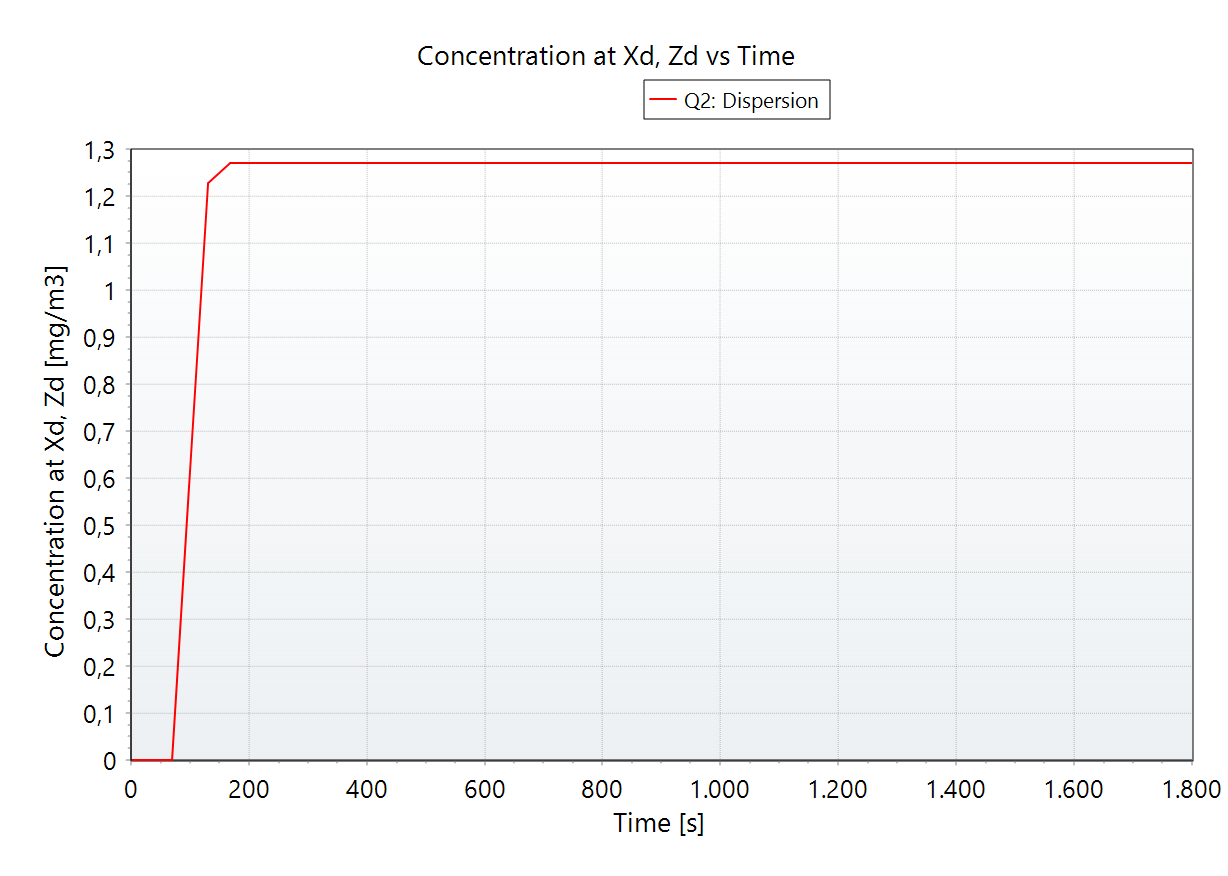 Toxic dispersion of HF - Concentration profile at a specific location. |
|---|
These functions can be also displayed at the same time (graph below) while the report displays exact values for specified height, time or downwind distance from the source.
.png)
Dose and Lethality over distance from toxic dispersion of HF
Conclusion
Battery Energy Storage Systems (BESS) present significant safety concerns, particularly in the event of thermal runaway. This study demonstrates the complexity of such incidents, highlighting the potential for prolonged toxic gas releases, including hydrogen fluoride, which poses serious health risks even at low concentrations. By modelling different BESS thermal runaway scenarios, we can better understand the associated hazards with toxic clouds. The analysis emphasizes the importance of robust safety measures and accurate modelling to mitigate risks and protect people and property.
Download the project file
Explore the project file simulating the BESS thermal runaway. Adjust map contours, and select different graphs or multiple graphs at once. Compare the results of different runaway scenarios and assess the damage effect.
To view the project file, please open it using the EFFECTS software. If you do not have the software, you can download and use the free viewing demo version of EFFECTS via the link below.


.png)